|
1/31/2024 0 Comments Examples of each dispersio forces
The phase in which a substance exists depends on the relative extents of its intermolecular forces (IMFs) and the kinetic energies (KE) of its molecules. The differences in the properties of a solid, liquid, or gas reflect the strengths of the attractive forces between the atoms, molecules, or ions that make up each phase. 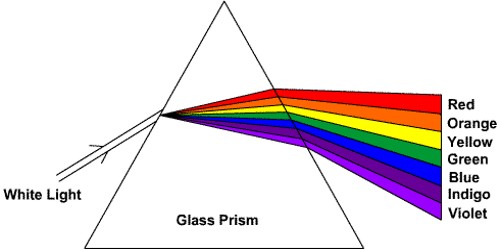
Particles in a solid vibrate about fixed positions and do not generally move in relation to one another in a liquid, they move past each other but remain in essentially constant contact in a gas, they move independently of one another except when they collide.Particles in a solid are tightly packed together and often arranged in a regular pattern in a liquid, they are close together with no regular arrangement in a gas, they are far apart with no regular arrangement.Note that we will use the popular phrase “intermolecular attraction” to refer to attractive forces between the particles of a substance, regardless of whether these particles are molecules, atoms, or ions.Ĭonsider these two aspects of the molecular-level environments in solid, liquid, and gaseous matter: In the following description, the term particle will be used to refer to an atom, molecule, or ion. Explain the relation between the intermolecular forces present within a substance and the temperatures associated with changes in its physical stateĪs was the case for gaseous substances, the kinetic molecular theory may be used to explain the behavior of solids and liquids.Identify the types of intermolecular forces experienced by specific molecules based on their structures. 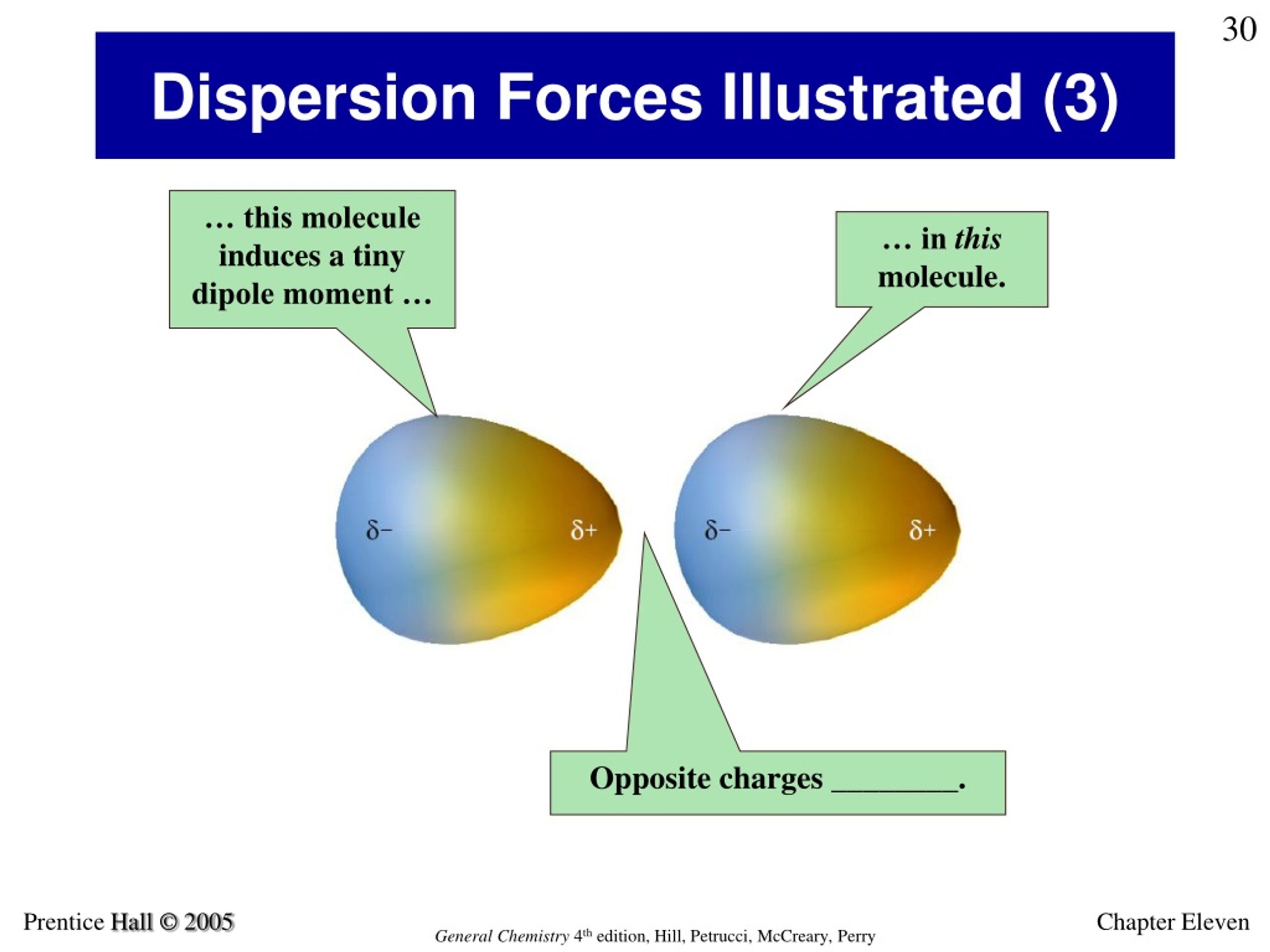
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
